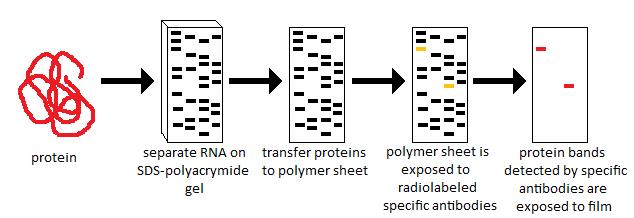
Western blotting is not only used for detecting specific proteins but also for quantifying their relative abundance. Proper quantification is essential for comparing protein expression across samples or experimental conditions. Several methods exist to quantify western blot signals, each with its advantages and limitations.
1. Densitometry
Densitometry is the most common method for western blot quantification. It measures the intensity of protein bands from an image, which is proportional to protein abundance.
Workflow:
-
Capture the blot image using a digital imaging system.
-
Use software (e.g., ImageJ, Bio-Rad Image Lab) to define the band area.
-
Measure the integrated density or pixel intensity.
-
Subtract background signal to correct for non-specific noise.
-
Normalize band intensity to a loading control (e.g., β-actin, GAPDH).
Pros: Simple, widely used, compatible with chemiluminescent or fluorescent detection.
Cons: Limited dynamic range for overexposed bands; careful normalization required.
2. Fluorescent Western Blot Quantification
Fluorescent western blotting uses secondary antibodies labeled with fluorescent dyes instead of enzymes. Detection is performed using fluorescence scanners.
Workflow:
-
Incubate the blot with fluorescently labeled secondary antibodies.
-
Scan using a fluorescence imaging system.
-
Measure fluorescence intensity of each band.
-
Normalize to internal loading controls or housekeeping proteins.
Pros: High sensitivity, wide dynamic range, allows multiplexing multiple proteins on the same blot.
Cons: Requires specialized imaging equipment; more expensive than chemiluminescence.
3. Chemiluminescence Quantification
In chemiluminescent detection, horseradish peroxidase (HRP) reacts with a substrate to emit light, which is captured using film or a digital imager.
Workflow:
-
Apply chemiluminescent substrate to the blot.
-
Capture the emitted light using X-ray film or a CCD camera.
-
Digitize the image and measure band intensity using densitometry software.
-
Normalize to loading controls.
Pros: High sensitivity and standard in most labs.
Cons: Limited linear range if bands are overexposed; film-based detection is less quantitative than digital systems.
4. Normalization Techniques
Proper normalization is critical for accurate quantification. Common methods include:
-
Housekeeping Proteins: Compare the target protein to a stable internal control like β-actin, GAPDH, or tubulin.
-
Total Protein Staining: Use stains like Ponceau S or Coomassie Blue to measure total protein in each lane, reducing variability caused by uneven loading.
-
Ratio Analysis: Express protein levels as a ratio of target protein intensity to control protein intensity for comparative analysis.
5. Advanced Quantification
Some labs use software for more advanced quantification, which may include:
-
Non-linear regression: For chemiluminescent signals to correct saturation.
-
Background subtraction algorithms: Improves accuracy in noisy images.
-
Multiplex fluorescence analysis: Measures multiple interpreting western blot results simultaneously with internal controls.
Tips for Reliable Quantification
-
Avoid overloading or underloading protein samples; both can distort quantification.
-
Ensure exposure time is within the linear detection range.
-
Always run replicates to account for biological variability.
-
Confirm the stability of the loading control under experimental conditions.
By combining careful experimental design with appropriate quantification methods, western blotting can provide not only qualitative but also robust quantitative data on protein expression and regulation.

